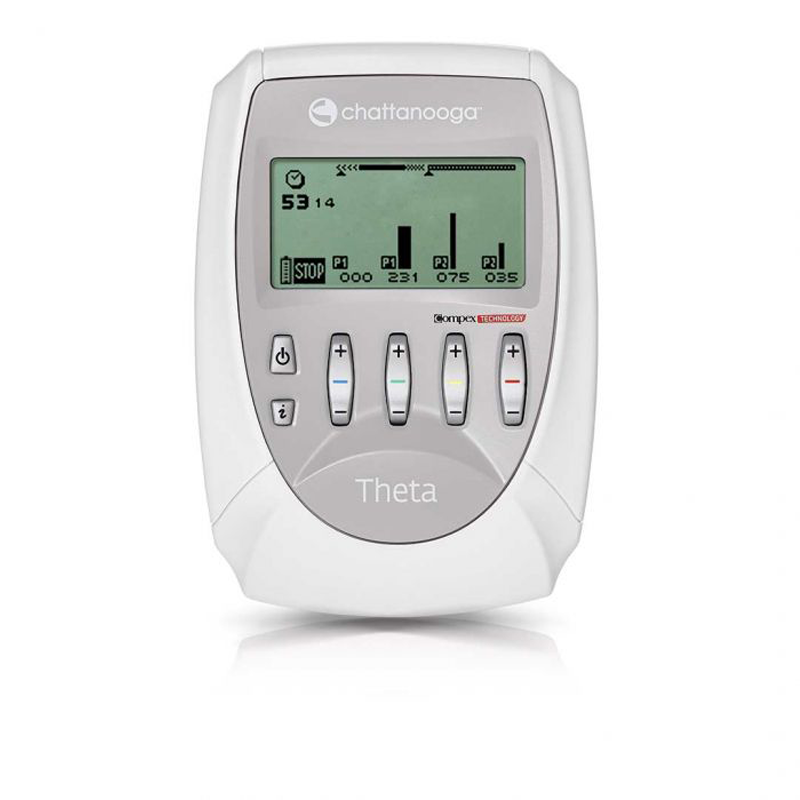
Chattanooga Theta Electrostimulation Device
799,00 €
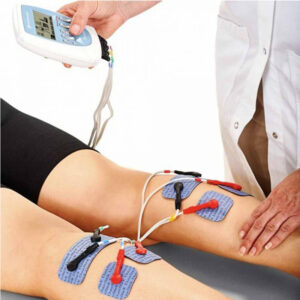
Chattanooga Theta electrostimulation device is used for electrotherapy. The device has four-channel stimulants and can handle standard and specific protocols. It is used by physiotherapists and rehabilitation clinics. The Chattanooga Theta electrostimulation device provides many different protocols. Theta electrostimulator has more options than the Rehab electrostimulator. Chattanooga Theta is an easy-to-use device.
The Chattanooga Theta electrostimulation device has four standard and four full programs, namely rehabilitation I and II, pain relief I and II, vascular, neurological rehabilitation, and conditioning program I and II. Compared to the Chattanooga Rehab, the Theta electrostimulator has many more options. It also has 2 Mi-cables “Muscle Intelligence”, which makes it suitable for using Muscle Intelligence Technology.
The Chattanooga Theta electrostimulation device has four channels. You can choose one app for all four channels, but you can also choose to turn on one app for the first two channels and choose another app for the third and fourth channels. In this way, it is possible to train several parts of the body at once. The electrostimulation device monitors the time of the session and automatically moves to the next stage when necessary.
The composition of the kit:
- 1 Chattanooga Theta device
- 1 Charger with EU plug
- 1 battery
- 1 pin cable set (4 pcs.)
- 1 Snap Adapter Kit
- 2 Mi-cables
- 1 Protective cap
- 2 Electrode sets 5x5cm (1 snap connection)
- 2 Electrode sets large 5×10 cm (2 snap connections)
- 1 instruction manual and practical guide on CD/USB
- 1 Quick Start Guide + Warning Brochure
- 1 Electrode placement brochure
- 1 Bottle of gel
- 1 Motor point pen
- 1 Transport bag
- 1 Belt clip
Programs:
- 4 standard + 4 Full programmes categories:
- Rehabilitation I&II
- Pain Relief I&II
- Vascular
- Neurologic Rehabilitation
- Conditioning I&II
It is possible to purchase this product by leasing. About leasing options, write to info@fiziopreces.lv |
 |
Aizkavētais pasūtījums





















Reviews
There are no reviews yet.